WARNING:
On 4/22/11, about a year after discontinuing the testicular heating started in November, 1998, I received
results of my semen analysis which indicated that the method is not completely reversible. The abnormalities were low motility (<1% rather than >60%), high
abnormal morphology (60% rather than <40%) and low count (16 million/mL rather than >19 million/mL). Until this result, every other report suggested that the
heating method was fully reversible. After an additional year (on 2/29/12) semen analysis still gave abnormal results: 26% motility, 65% abnormal
morphology and a count of 14 million/mL. Another test on 8/20/14 from Quest Diagnostics, about four years and four months after discontinuing 11 years of heating, showed little
improvement: 30% motility, a count of 22.5 million/mL and an unspecified but abnormal percentage of abnormal morphology.
On 12/3/14 a semen analysis by the usual Sutter General Hospital lab found a motility of 25.6%, 60% abnormal morphology
and a count of 12 million/mL. This test was after 14 weeks of daily oral dosing with 250 mg of ursolic acid to see if it
had any effect on sperm motility, which it apparently did not, so the dosing was discontinued.
The probability of conception can be made very small by sufficiently reducing the amount of sperm a man produces. This reduction in sperm production is easily accomplished by bringing the temperature of the testicles closer to body temperature, and this is most conveniently done by bringing the testicles closer to the body. Preliminary studies show that this method is effective: there were no pregnancies for the nine couples over 151 ovulation cycles of exposure.
My wife and I had considered every conventional method of birth control and we were very frustrated with their shortcomings. The artificial hormones like birth control pills made her feel depressed, spermicide on condoms burned her skin and were too ineffective without it, tuboligation and vasectomy seemed premature because we weren't certain we wouldn't want a child later, and a diaphragm was too difficult to install and not very effective without spermicide anyway. For a time it seemed that the symto-thermal method of predicting fertility would work. For several months we charted my wife's menstrual cycle. The main problem was that we had to abstain when her libido was the highest - the fertile portion of her cycle! This inspired me to get on the net and into the library and find another method.
The testicular heating method has been ideal for us, but we are an unusual couple. The advantages are that it doesn't cost anything after getting it started, it takes little time to maintain once started, it doesn't limit sexual activity in any way, it doesn't require exposure to or consumption of any drugs or hormones, and it is reversible. The disadvantages are that it hasn't been tested by many couples, so there is an unknown risk to the health of the testicles and to any baby resulting from a failure of the method. With each new study the risk gets smaller. Another disadvantage is that there is little support for this method. Most people don't even know about it!
One concern that this method brings to mind is the association of raised testicles to testicular cancer, and the best studied variation of this method involves raising the testicles. In my work as a toxicologist I attended the 40th annual meeting of the Society of Toxicology in San Francisco in March 2001. One of the first speakers was N. E. Skakkebaek of the Copenhagen University Hospital in Denmark. In his presentation entitled, "Human Testicular Cancer: Secular Trends and Fetal Origin," he described how the impaired development resulting from raised testicles in infants results in testicular cancer in adolescence. When I asked him after the session whether raised testicles could lead to cancer in adults, he replied that there was no reason to expect so, since the testicles of an adult are already developed.
This brings up the question: If this method is so simple and effective, why isn't it well known and used? Probably because there isn't much demand for it. Most people are satisfied with existing methods. Since the only thing needed to make this method work is a rubber band, there is limited profitability in promoting or marketing the needed supplies. Condoms make much more money, and those big money makers would have to be competed against to introduce a new birth control method for men.
First, I read the available research thoroughly. Like the researchers, I set as my goal to get my sperm count consistently below one million sperm per milliliter. Before changing my sperm production, though, I needed to determine what my sperm production was already, and establish a method to measure it. Fortunately I had a microscope already, capable of magnifying 400 times, and could see sperm using it.
Measuring sperm concentrations using my microscope posed some challenges. A sample on a slide would show about one hundred squirming cells moving in and out of the viewing area. I needed to immobilize the cells to make counting possible, but without changing their concentration uncontrollably. Then I needed a way to calculate the sperm concentration in the sample from the number of sperm counted in the viewing area.
To complete this method there were two pieces of information still needed: What is the total area occupied by the sample, and how large an area is visible through my microscope at a magnification of 400? The first question is easily, given a few assumptions. The cover glass for my slide measures 1.8 cm by 1.8 cm, and five microliters of sample is sufficient to fill this area without any excess oozing out from under the slide cover. Assuming that the sample applied to the slide is representative of the sample as a whole, and that the sample, and sperm, will be evenly and reproducibly distributed throughout the area of the slide cover, then it is possible to calculate the sperm concentration in the original sample from the number of sperm seen in a small, known area of the slide.
The second question was more tricky. To answer it I needed to know the area visible through the microscope at a magnification of 400. I approximated this by noting that a nichrome wire, with a measured thickness of 0.5 mm using a caliper, was slightly wider than the visual field when viewed under the microscope. Therefore I estimated the diameter of the visual field to be 0.4 mm, for a total area of 3.14 * (0.2 mm)2 = 0.13 mm2. Since the area of the slide cover is 18 mm * 18 mm, or 324 mm2, the portion of the slide cover area which can be viewed by the microscope at one time is 324 mm2/0.13 mm2 = 1/2578.
Preparing a sample for examination was done as follows: A sample of semen obtained after three days of abstinence was placed in a glass vial, weighed, and set aside for 15-20 minutes, by which time the clumpiness has disappeared and the sample is easily mixed to give a homogeneous suspension. Five microliters of 37% formaldehyde was injected from a glass syringe, followed by a drop of saturated aqueous gentian violet. The sample was stirred, drawn into the cleaned syringe, and five microliters of it were placed on a microscope slide. The slide was examined at 400 magnification in 20 different places to minimize any unevenness in the distribution of sperm, and the concentration of sperm in the original sample was calculated from the average number of sperm in each viewing area according to the following sample calculation:
Notice the correction in the above calculation for the slight dilution
of the original sample by formaldehyde and gentian violet solution.
There is also the assumption that the density of semen is 1.000 g/mL,
which is surely low by several percent.
After testicular heating reduced the concentration of sperm in the
sample, taking the measurements became easier. Since there was usually
less than one sperm in each viewing area, there was no need to
immobilize them with formaldehyde. By forgoing the stain with gentian
violet, it became possible to place the semen sample directly on the
microscope slide. Using a syringe to measure the volume of semen being
placed on the slide was problematic because the needle often clogged.
Given that the measurement being sought is the order of magnitude of
the sperm concentration, the sample can be applied to the slide
directly. If significantly more than five microliters is used, it will
ooze out from under the slide cover. If significantly less is used, it
won't fill the area under the cover. Whether the depth of the sample
varies in these cases is an area needing further study.
Although the original article used special
underwear equipped with a hole and rubber band to keep the testicles
suspended in the abdomen, I found that a rubber band around the base of
the penis, as close to the abdomen as possible, is all that is needed.
The penis and empty scrotum are on one side, and the testicles in the
abdomen are on the other. The rubber band should be just tight enough
so that the testicles can't slip past it and return to the scrotum, and
it should be loose enough to leave circulation unimpaired. A wide
rubber band is preferable because it is less likely to roll away from
the abdomen.
The best rubber band I found for this purpose is the kind used to
bundle stalks of broccoli at the supermarket. I put it on after my
shower every morning, and remove it before bed each night. Between uses
I clean it with soap and water. During the first months of wearing it I
checked periodically during the day to make sure that my testicles were
still suspended. It became less and less likely that I would find that
my testicles had left my abdomen, perhaps because the space they filled
had adjusted to them. Soon I didn't need to check any more because my
testicles usually remained in my abdomen until the rubber band was
removed, and I learned to feel when they left. At no time was it
difficult to release my testicles back to the scrotum. After six months
the rubber band had become too loose and needed to be replaced.
Although the rubber band has generally been perfectly comfortable, even
unnoticeable, it can lead to some itching, especially at the end of the
day. Since latex tends to produce irritation, a different material such
as elastic or silicone rubber may improve this, but these haven't been
tried because the problem has been so minor.
However, after the first three years (late 2002) the irritation
increased, and it became necessary to find a less irritating material.
What I ended up using is a loop of yarn which is replaced daily. The
loop goes around my penis at the base and is secured to my underwear in
the part between the legs by a safety pin.
This method is based on the observation that slight pressure at either
side of the pelvis would prevent the testicles from descending. The
purpose of the safety pin is to prevent pressure on the bottom of the
scrotum, which seems to be the most sensitive part, and to give some
elastic tension to the system. This method significantly reduced
itching, but takes more adjustment to make it effective than the rubber
band did. For several months I simply tied the ends of the yarn above
the base of my penis to make the loop snug, but this tended to become
too tight or too loose within the same day and couldn't be easily
adjusted. Recently I purchased a "cord stop" from a fabric store, a
spring loaded cylinder for securing cord ends, commonly seen on
backpacks, and this allows the loop of yarn to be adjusted.
One complication throughout this time which I haven't mentioned is that
physical activity, especially kneeling or crawling, increases the
likelihood that the restraint system will fail. This hasn't been a major
problem for me because most of my day isn't very active. If it were a
problem, I would test whether using the restraint system while sleeping
would be effective.
Here is a description of how I install this yarn:
To verify the accuracy of the sperm concentration measurement method I
had developed, an analysis of my sperm concentration was made by an
independent laboratory before and after the testicular heating method
was begun. Before testicular heating my sperm count was normal, and 43
days after beginning the heating I was found to be infertile. The
measurement of 400,000 sperm/mL was based on the observation of seven
sperm on the analysis slide. Since they were all dead sperm, the likely
concentration of live sperm is much lower. These results are comparable
to the measurements made using my own microscope.
The table below shows my sperm count before and after testicular
heating was started. All measurements were made personally except where
indicated otherwise.
A few people have tried to use this method but have reported difficulty telling
whether thay are using it correctly. This was the preliminary advice I sent:
UPDATE: On 4/12/11, about one year after discontinuing the heating, I had the opportunity for another testosterone test at the same lab as
previously. This time the total testosterone was 319 ng/dL and the free testosterone was 56.8 pg/mL, with a % free testosterone of (5.68 ng/dL / 319 ng/dL) *
100% = 1.78, all now within the reference ranges.
Here is a comment I received from a friend who has started using this method:
Date: Thu, 11 May 2006 19:14:20 +0200
Hi Chris
We had some correspondence regarding the testicular heating. And my question was at some point
whether it would influence testosterone levels. I had it checked and my level was normal.
System still working great.
Cheers
Anthony
You are welcome to contact
me for more information about this birth control method. If you are
considering attempting it yourself I would be happy to help you design
you own analysis procedure and interpret the data from it. This method
is experimental and requires great care. It should only be considered
by a couple who can afford to have the method fail. Only a handful of
couples have used this method - there is no guarantee that it will work
equally well for everyone. That said, I also think that this method is
drastically underused.
************************************
Here is a brief conversation from someone who developed a thermally insulated cup to accomplish the heating:
On Sun, 15 Nov 2015:
Hi,
On Wed, Nov 18, 2015 I wrote:
Thanks.
It might work. The important thing is to make a standard method and then show that it effectively and consistently reduces sperm count
to infertile levels.
On Thurs, Sep 2, 2021:
Chris,
This took a very long time for me to reply, but I wanted to let you know that I have been successful as well.
Thanks again.
On Fri, Sep 3, 2021 I wrote:
Thank you for the long-term follow-up! Can you confirm that the cup is only providing thermal insulation, and isn't causing your testicles
to be pushed into the abdomen?
Yours,
On Fri, Sep 3, 2021:
Chris,
It is doing both. Thermal and with help of 'speedo' underwear push testicles up.
On Mon, Sep 6, 2021:
Chris,
I read everywhere online that to increase sperm, wear loose-fitting, so I did the opposite.
As for these thermo materials,
they don't seem to sell in US anymore, but I cut my cups out of these:
Thanks again.
************************************
A few relevant links:
Heat based contraception - natural birth control for men?
Male Contraception Information Project - Heat Methods
Ultrasound male contraceptive, overlooked for decades, confirmed to work - EurekAlert! PDF

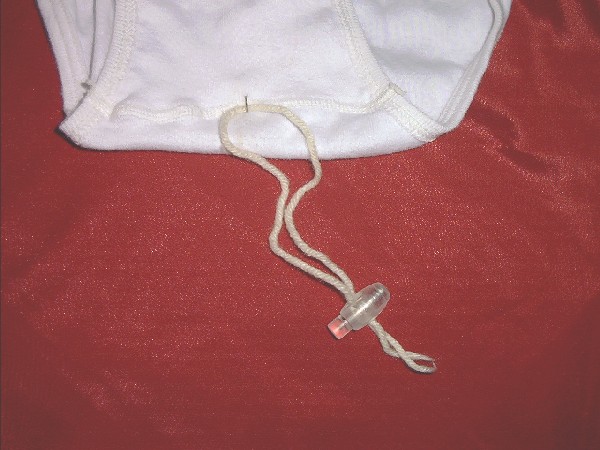

My morning routine
is to snap off about a foot of yarn and tease the middle of it through the
opening of a cord stop using the tip of a safety pin. The piece of yarn
forms a loop with the cord stop clamping the ends. Then I use the safety
pin to attach the loop to the inside of my underwear, at the point midway
between the leg holes where there is a seam. Then, after I put on the
underwear, I move my testicles into my abdomen and put my penis and
scrotum through the loop of yarn, with the cord stop at the top of the
loop. Then I slide the cord stop to tighten the loop sufficiently to keep
my testicles in place without interfering with circulation. During the day
I may need to slide the cord stop again to take up slack that forms,
especially if I have been physically active, and often I have to return my
testicles to my abdomen. I recently discovered that I could prevent this
slippage by tying knots in both tails of the yarn at the point where they
come out of the cord stop.
Date
Days from Start of Heating
Live Sperm/mL
11/17/98
-6
39 million
11/20/98
-3
57 million
11/23/98
0
89 million*
11/30/98
7
21 million
12/3/98
10
2300 thousand
12/7/98
14
57 million**
12/10/98
17
54 million
12/15/98
22
26 million
12/18/98
25
32 million
12/21/98
28
7000 thousand
1/5/99
43
400 thousand*
1/8/99
46
<26 thousand
1/12/99
50
<26 thousand
1/15/99
53
<26 thousand
1/20/99
58
<26 thousand
1/29/99
67
<26 thousand
3/3/99
100
<26 thousand
4/1/99
129
<26 thousand
9/30/99
311
280 thousand
12/10/99
382
130 thousand
2/28/00
462
26 thousand
1/8/01
777
490 thousand
8/8/01
989
77 thousand
3/28/02
1221
<26 thousand
5/2/03
1621
<26 thousand
4/23/04
1978
26 thousand
6/14/07
3125
4 million!***
7/3/07
3144
335 thousand
9/24/07
3227
<26 thousand
3/25/10
4140
<26 thousand
5/6/10
4182
<26 thousand****
7/8/10
4245
10 million*****
9/2/10
4301
21 million*****
4/20/11
4531
16 million*
2/29/12
4846
14 million*, ******
8/20/14
5749
22.5 million#
12/3/14
5854
12 million*,##
* These four measurements were made by the Medical Center of the
University of California at Davis through Sutter General Hospital in Sacramento
** This semen sample was obtained after four days of abstinance
rather than three.
*** On 6/14/07 I started using this method 24 hours/day, rather than
during the daytime only (~16 hours/day) as I had previously
**** The heating method was discontinued on this day (5/6/10). I was 42 years old.
***** 7/8/10: I am guessing there are 20 live sperm per visual field - too many for an exact count, but definitly within 10-30.
9/2/10: I am guessing there are 40 live sperm per visual field - too many for an exact count. Diluting the sample didn't give any
visible sperm.
****** This semen sample was obtained after two days of abstinance
rather than three.
# This test was performed by Quest Diagnostic in Sacramento, CA
## This was measured after 14 weeks of daily 250 mg oral ursolic acid to determine the effect on motility.
I'm still using the method I posted, but you'll notice that I changed my
method from using a rubber band to using a piece of yarn to reduce skin
irritation from the rubber. There are two things you need to check if you
want to use this method with confidence. The first is that your testicles
are kept out of your scrotum for most of the day, at least. This can be
achieved with a rubber band or a piece of string or yarn. It doesn't
matter which method you use as long as your testicles are kept out of your
scrotum most of the time. The place the rubber band fits is around the
base of the penis with the penis and empty scrotum on one side of the
rubber band and the testicles and the rest of your body on the other.
You can make sure that you are doing the method correctly by having your
sperm count measured both before and after you begin using the method.
This measurement can be provided by a medical laboratory or you can make
it yourself if you are confident with a microscope and the mathematical
procedure I described on puzzlepiece.org. Two measurements showing
fertility before starting the method (>~100 million sperm/mL) and
infertility after using the method for a month or two (~<1 million
sperm/mL) will confirm that you are using the method correctly, and that
the method is working despite any adjustments you have made to it.
I don't have pictures at the moment, but your body may work differently
from mine. I found the rubber band from a batch of broccoli to be about
the right size for me, at least for broccoli purchased in the United
States. When you stand up, the skin in the pubic area tightens, so this is
when the testicles are more likely to slip past the rubber band and
descend back into the scrotum. I have this problem when the rubber band is
too large. There are a range of tightnesses that prevent the testicles
from getting past the rubber band without interfering with bloodflow.
Maybe the rubber band won't work for you if your body is much different
than mine, or if you are more physically active than I am as a programmer.
In this case you might experiment with using the method at night instead
of the daytime - but you would need to check your sperm count to confirm
that the method works with only overnight heating (~8 hours) rather than
daytime heating (~16 hours).
UPDATE: After eight years of continuous daytime use, I found that my sperm count
had begun to recover to unacceptably high levels (see above). Perhaps there is
some kind of tolerance that occurs over the years. I started using the method at night
also (i.e., continuously, 24 hours/day) and my sperm count seems to have dropped
back to zero.
UPDATE: On 4/27/10, in anticipation of discontinuing the heating, I had serum testosterone measured as part of the serum analysis during routine physical
examination. The physical examination and serum analysis didn't reveal anything unusual (except that lymphocytes as percent of platelets was 22%, below the
reference range of 26.0% - 46.0%), and the total testosterone level was 487 ng/dL, well within the reference range of 250 to 1100 ng/dL. The "free testosterone"
was 61.7 pg/mL, also within the reference range of 35.0 to 155.0 pg/mL. Strange that they changed the units. Then they calculated the "% free testosterone"
which is just the quotient of these two numbers, accounting for units, where 61.7 pg/mL = 6.17 ng/dL, so the % free testosterone is (6.17 ng/dL / 487 ng/dL) *
100% = 1.27, where the reference range is 1.5 to 2.2. I will have to study up and consult my doctor to see if this abnormality has any meaning. It appears that
a larger proportion of my testosterone is bound to something, a protein maybe, than for most other men my age, although testosterone production has been normal.
I plan to duscuss these results in August, assuming that my sperm count appears normal, when I ask my doctor for a referral for a sperm count
measurement.
UPDATE: It appears that two months after discontinuing the heating (which I practiced diligently for over 11 years) I am now fertile
(10,000,000 sperm/mL > 1,000,000 sperm/mL) and recovering.
UPDATE: On 12/13/14, after failure of ursolic acid to affect sperm motility and needing contraception, I started using
the heating method again 24/7.
UPDATE: On 10/22/15 I stopped using the yarn 24 hours/day and went back to using it only at night. Given the low fertility of my wife and I, I decided it
wasn't worth the hassle of adjusting the yarn and testicles all day for what was probably an unnecessary margin of effectiveness.
WARNING:
On 4/22/11, about a year after discontinuing the testicular heating started in November, 1998, I received
results of my semen analysis which indicated that the method is not completely reversible. The abnormalities were low motility (<1% rather than >60%), high
abnormal morphology (60% rather than <40%) and low count (16 million/mL rather than >19 million/mL). Until this result, every other report suggested that the
heating method was fully reversible. After an additional year (on 2/29/12) semen analysis still gave abnormal results: 26% motility, 65% abnormal
morphology and a count of 14 million/mL. Another test on 8/20/14 from Quest Diagnostics, about four years and four months after discontinuing 11 years of heating, showed little
improvement: 30% motility, a count of 22.5 million/mL and an unspecified but abnormal percentage of abnormal morphology.
On 12/3/14 a semen analysis by the usual Sutter General Hospital lab found a motility of 25.6%, 60% abnormal morphology
and a count of 12 million/mL. This test was after 14 weeks of daily oral dosing with 250 mg of ursolic acid to see if it
had any effect on sperm motility, which it apparently did not, so the dosing was discontinued.
From: Anthony
To: Chris Jenks
Subject: Re: Male contraceptive
Thanks for your your blog.
What's you opinion about using protective cups as another form of birth control?
I know it gets pretty warm in a cup as well.
Thanks.
I made a soft cup (breathable wet suit material) in tight-fitting underwear. Much more comfortable.
I have been able to bring my sperm count to zero. Nothing is seen in a microscope.
Really amazing nothing like this exists already.
Of course, I do not know the long terms effects afterward if I stop, but I'm fine with that.
I would like to post your observation (anonymously) on my website, puzzlepiece.org.
Chris
Yes, please go ahead and post this on your website. I can send some pictures and more info. later.
I found these underwear/swim briefs that were tight
(I actually got one size smaller too).
It worked out perfectly because it had a liner inside that acted like a pocket so I could slip my thermo cups inside like a jock.
My body temp vs temp down there is 98.3, basically the same.
Hope this helps your readers.
If they need any help in cutting the cups, let me know.